Ticagrelor and ASA vs. ASA Only After Isolated Coronary Artery Bypass Grafting in Patients With Acute Coronary Syndrome (TACSI)

Study design
Interventional, multi-national, multi-center, prospective, randomized, open, blinded end-point registry-based clinical trial.
About the project
Patients with acute coronary syndrome undergoing coronary artery bypass grafting are postoperatively 1:1 randomized to receive therapy with ASA alone or ASA and ticagrelor for 1 year. In total, 2200 patients from twenty Nordic cardiac surgery centers will be included.
The primary endpoint is a combination of death, myocardial infarction, stroke and new revascularization during the first 12 months after randomization. Secondary endpoints are: individual major adverse cardiac events (MACE) elements, cardiovascular hospitalization, bleeding, dyspnoea, renal failure, atrial fibrillation, and MACE at 2, 3, 5 and 10 years.
Objective(s)
To compare single antiplatelet therapy with acetylsalicylic acid (ASA) or dual antiplatelet therapy with ASA and ticagrelor after coronary artery bypass grafting in patients with acute coronary syndrome.
Collaboration
22 participating centres: Sweden (all 8), Denmark (all 4), Norway (4), Finnland (5), Iceland (1).
Project group
- Anders Jeppsson (Sponsor), Sahlgrenska University Hospital, Gothenborg, Sweden
- Ivy Susanne Modrau (institutional principal investigator), Aarhus University Hospital, Aarhus, Denmark
Funding
The Swedish Research Council (Vetenskapsrådet) and the Swedish Heart-Lung Foundation.
Published papers
- Björklund E., Malm C. J., Nielsen S. J., Hansson E. C, Tygesen H., Romlin B. S., Martinsson A., Omerovic E., Pivodic A., Jeppsson A. Comparison of Midterm Outcomes Associated With Aspirin and Ticagrelor vs Aspirin Monotherapy After Coronary Artery Bypass Grafting for Acute Coronary Syndrome. JAMA Netw Open. 2021;4(8):e2122597. DOI: 10.1001/jamanetworkopen.2021.22597
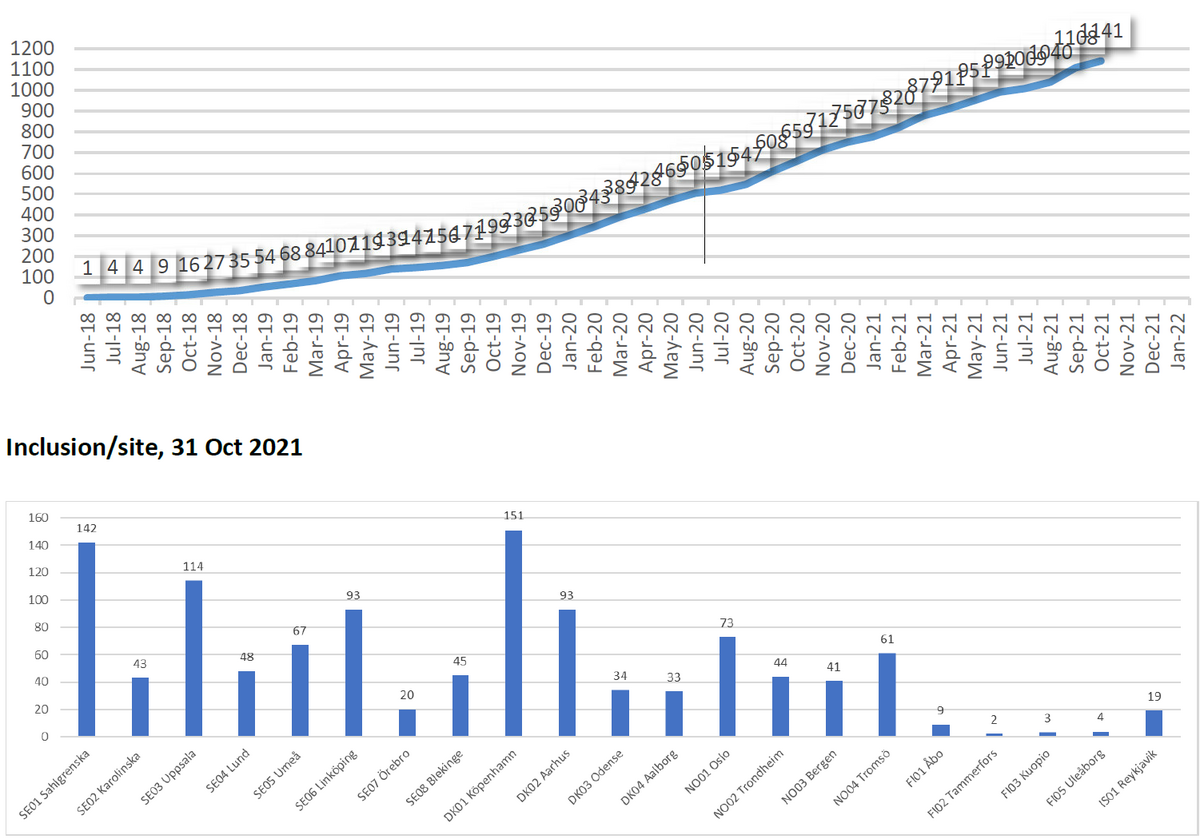
Status
Inclusion ongoing
Project period
Start: 1 June 2018
End: 1 December 2024
EudraCT number
2017-001499-43
