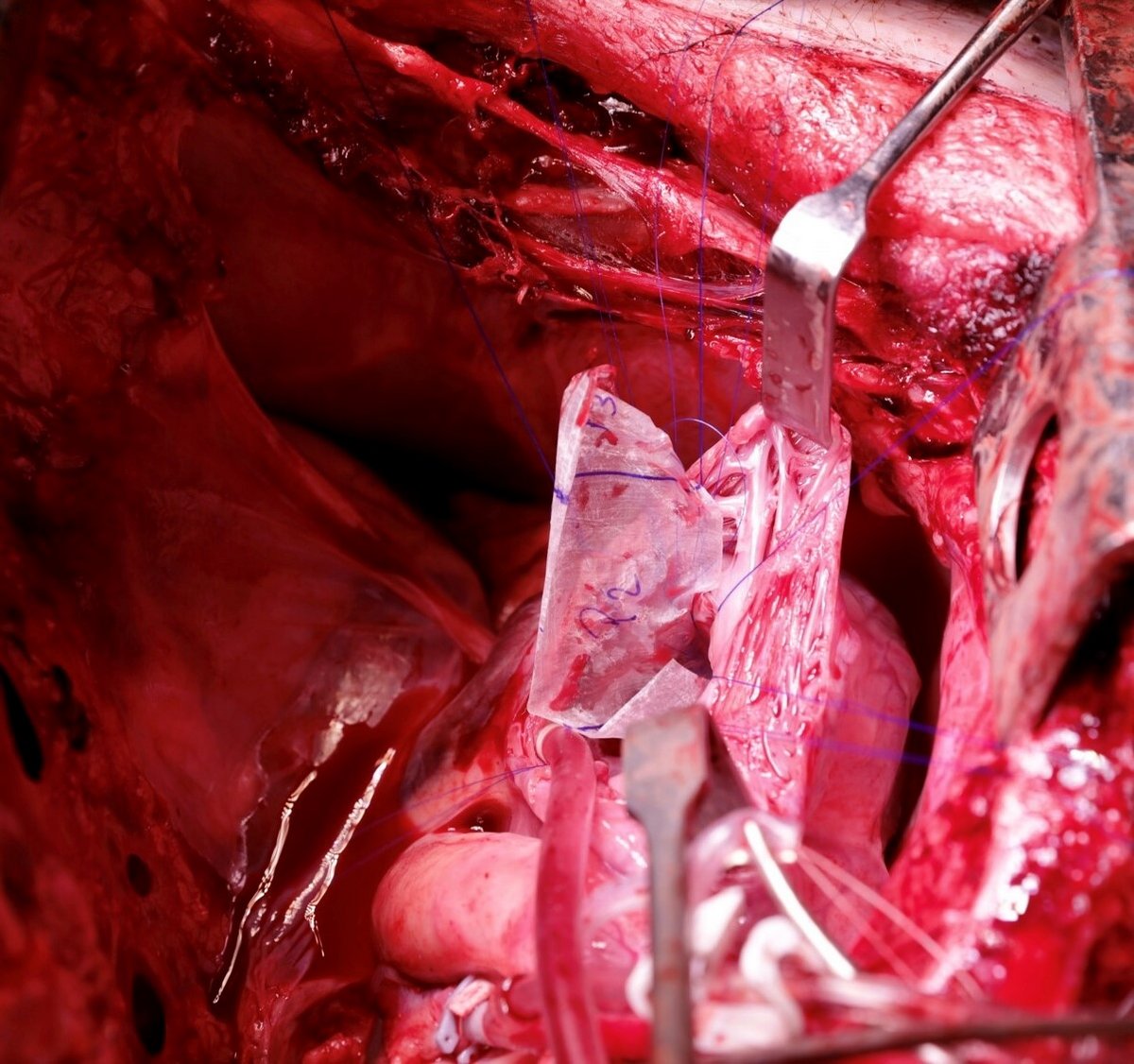
Reconstruction of the Entire Posterior Mitral Valve Leaflet Using Small Intestinal Submucosa Extracellular Matrix
Study design
Experimental porcine study.
About the project
Regurgitation is the most common disease in the mitral valve. As a result, mitral valve repair surgery procedures have increased significantly over the past five decades. In either type of mitral valve disease, it is widely accepted that mitral valve repair has an advantage over mitral valve replacement. However, severe cases of degenerative valve disease may necessitate an entire mitral valve replacement in the long term.
By refining the current surgical treatment options for heart valve disease and developing new ones, we can ultimately provide better, sustained care for patients suffering from heart valve disease. This study in reconstructive mitral valve surgery with bioscaffold materials has the potential and perspective to provide a better and alternative treatment option for many patients - especially patients with severe pathology that would otherwise only be eligible for mitral valve replacement, which has proven subpar compared to mitral valve repair. If our method proves successful, we can offer mitral valve repair for all patients with mitral regurgitation, regardless of pathology. Furthermore, no artificial material will be left in the operated area, which will significantly lower complications associated with heart valve surgery, such as the need for life-long blood-thinner therapy, risk of endocarditis, as well as the potential need for reoperation.
Objective(s)
The purpose of this study is to investigate new bioscaffolds for heart valve repair and replacement - both in vitro and in vivo - to validate biomechanical performance and conceive new surgical concepts in mitral valve surgery.
Collaboration
The project is anchored at Cardiothoracic and Vascular Surgery, Department of Clinical Medicine, Aarhus University Hospital / Aarhus University. Locally collaborative partners include the Department of Histopathology and the MR Centre, Aarhus University Hospital. Internationally, we collaborate with CorMatrix, Cardiovascular Inc., Alpharetta, GA, USA.
Project group
- Johannes H. Jedrzejczyk, BSc, Medical Student, PhD Student
- Marcell J. Tjørnild, MD, PhD Student
- Søren N. Skov, MSc, PhD
- J. Michael Hasenkam, MD, DMSc, Professor
Funding
The project has received funding from Novo Nordisk Fonden and Grosserer A.V. Lykfeldts og Hustrus Legat.
Status
Ongoing
Project period
Start: 1 January 2021
End: 1 July 2025
