Patient cohorts
The role of our clinical studies
Our aim is to carry out the most detailed studies of people with Parkinson's Disease that has ever been done. We include large groups of patients in the early disease stages and study them with different scanning techniques, clinical measurements, sleep studies, mobile phone apps, and other methods. Our patients are then followed for years to study the variable disease development among different patients.
Current projects
PET studies of patients with prodromal and de novo Parkinson's disease - cohort 1 (The PACE-I cohort)
Introduction
Parkinson's disease (PD) is characterized by protein aggregates (including Lewy bodies and Lewy neurites), but their origin and route of spread remains unclear. Recent research indicates that many PD patients develop symptoms from the autonomic nervous system and lose their sense of smell several years before the diagnosis. Lewy bodies and neurites have also been detected in the gut nervous system up to 20 years before the diagnosis. Furthermore, the gut bacteria composition is altered in PD patients. Based on these findings we hypothesise that PD may begin in the peripheral autonomic nerve endings and spread via nerves to the central nervous system.
We have previously demonstrated that patients with REM sleep behaviour disorder (RBD), an early symptom of PD, have reduced nerve supply to internal organs (gastrointestinal tract and heart), comparable to some diagnosed PD patients. However, their brain dopamine system is only mildly affected, and they have no motor symptoms. This suggests that the disease process in the periphery is present even in the early stages, supporting the hypothesis. Nevertheless, other studies have presented results in conflict with the theory of disease onset in the gut.
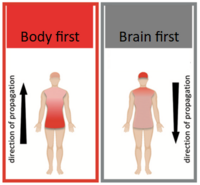
We propose two types of PD: body-first, starting in the intestine, and brain-first, where the pathology begins in the olfactory nerve. In a small percentage of patients (perhaps 10-15%), the disease may start simultaneously in the intestine and nose or spontaneously arise deeper in the brain.
Approximately 1/3 of PD patients develop RBD several years before receiving a PD diagnosis. According to our hypothesis, this strongly indicates a body-first type. The remaining 2/3 of patients without RBD before the PD diagnosis most likely have the brain-first type.
Purpose
The main objective is to examine newly diagnosed and untreated PD patients with and without the sleep disorder RBD and follow these patients over many years. This provides knowledge about differences in the development of the disease in patients with body-first PD and brain-first PD.
We perform detailed scans of the two patient groups to assess nerve system damage and its progression over time.
We also collect urine, blood, and stool samples to investigate factors that may initiate the characteristic tissue changes, and potentially contribute to PD development.
The aim is to understand how and where PD begins and how the pathology spreads, so that when treatments to halt or stop the progression of PD are available, we know which subtypes will benefit the most from different treatments.
Investigations
Sleep study
In this study, we examine PD patients shortly after diagnosis and patients without a PD diagnosis, but with very early signs of PD. All patients are followed over time, with examinations repeated every three years, allowing us to determine if the disease progresses differently depending on the subtype. Each trial period requires an overnight stay at a nearby hotel and participation in 2-4 trial days.
Polysomnography is a sleep study where a computer records brain electrical activity, eye movements, muscle activity, breathing, blood oxygen levels, and heart rate during sleep. Furthermore, a video camera records movement throughout the night. Thus, polysomnography can diagnose REM sleep behaviour disorder (RBD). Participation in the study is open to individuals with or without RBD.
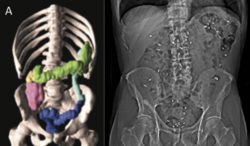
PET/CT scanning of the abdomen
Participants are injected with a very small amount of tracer substance [11C]donepezil, which is distributed in the body. This is followed by a PET scan of the abdomen, including a contrast-enhanced CT scan. The tracer is taken up by the so-called cholinergic nerve endings, providing a measure of the density of the parasympathetic nervous system in the intestines. This is an important measure, as the parasympathetic nervous system can be involved very early in body-first PD.
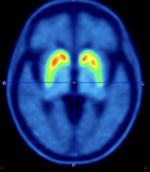
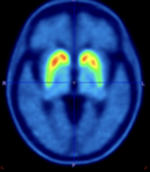
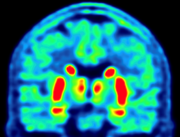
PET/CT scanning of the brain
Participants receive an injection with a small dose of the tracer [18F]PE2I. Subsequently, a PET/CT scan of the head is performed. The tracer measures the amount of functioning dopamine cells in the brain. The dopamine system is damaged in all patients with PD, but the symmetry and the progression of the damage might be different depending on the subtype of PD.
MRI scanning of the brain
During magnetic resonance imaging (MRI), multiple sequences are conducted including a T1-weighted anatomical sequence, a short-duration T2-FLAIR sequence to rule out other brain diseases, and a special neuromelanin sequence to measure the amount of neuromelanin in the brainstem. MRI is used to enhance the analysis of PET data and to measure certain brain systems that cannot be examined with PET scans.
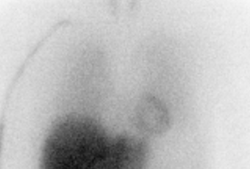
SPECT/CT scanning of the heart
Participants receive an injection of a small dose of tracer [123I]-MIBG, which visualizes the supply of the sympathetic nerve endings to the heart. Exactly 15 minutes later, an early static image of the heart is taken with a gamma camera. Approximately 3.5 hours later, another static image and a dynamic SPECT/CT recording are acquired.
This is an important measure as the sympathetic nervous system can be involved very early in some types of PD.
CT scanning of the abdomen
Every morning for 6 days a capsule containing 10 very small plastic markers, which the intestine cannot absorb, is ingested. The number of remaining markers in the intestine on the day of the scan can be converted into transit time through the gastrointestinal tract. This is thus a measure of intestinal function and particularly intestinal peristalsis (intestinal movements). Likewise, on this scan, the volume of the colon can be analysed as another measure of intestinal function and constipation.
Smell test
Participants are presented with 16 pens containing various familiar scents. From 4 options, they must identify the respective scent. The number of correctly identified scents is a measure of normal or impaired sense of smell.
Patients with PD often develop impaired sense of smell at various stages of the disease, making this an important test.
Biological samples
In addition to scans, we collect blood samples, a urine sample, and stool samples, which are analysed for protein aggregates, various inflammation markers and bacterial composition.
These samples are stored in a biobank for future research aimed at developing diagnostic markers, such as diagnosing PD through a blood test.
Intestinal permeability test
To measure the leakiness of the gut, participants ingest a solution containing 5 different types of sugars. The sugar molecules vary in size, affecting their absorption through the gastrointestinal tract. The amount of sugar excreted in the urine is a measure of gastrointestinal permeability.
This is an important test for obtaining deeper insights into how PD may potentially initiate in the gut's nervous system.
Blood pressure
Blood pressure is measured both in lying and standing positions to examine whether blood pressure drops more than usual (orthostatic hypotension). This is a symptom experienced by some patients with PD.
Motor symptoms
The motor symptoms are assessed using a standardized test (UPDRS), which evaluates the degree of slowness, stiffness, and tremors, among other factors. Motor function is also evaluated with more quantitative tests, such as moving the index finger back and forth between two points as many times as possible within one minute.
Other examinations
In addition to the above, a memory test is conducted along with various questionnaires covering symptoms commonly experienced with PD, such as constipation, sleep problems, etc.
Preliminary results

In 2020, we published the first results of our study. We demonstrated that PD (Parkinson’s disease) patients can be categorized into at least two subtypes:
- A "body-first" type, where early damage to the peripheral nervous system in the gut and heart is observed before the brain is affected, presumably because Parkinson's pathology starts in the peripheral nervous system and then spreads upward to the central nervous system.
- A "brain-first" type, where the pathology first appears and damages the brain before it spreads to the peripheral nervous system in the heart and gut.
It is very likely that the cause, the risk factors, the symptom burden, and the rate at which the disease progresses differ for these two types.
Horsager et al., Brain 2020; vol. 143, 3077-3088
The presence of RBD (REM sleep behaviour disorder) before motor symptoms is a marker of the body-first subtype.
Currently, we are validating these findings in a group of newly diagnosed PD patients, as well as conducting the first three-year follow-up study to demonstrate the robustness of the findings and to follow the progression of the disease in the two subtypes. Furthermore, we will start conducting a six-year follow-up study at the end of 2024.
The figure displays dopamine signal in the brain on the x-axis and sympathetic signal in the heart on the y-axis. Patients in the body-first group with RBD (blue dots) initially loose MIBG signal, followed later by loss of dopamine signal, and then eventually convert to PD with RBD. Conversely, PD patients in the brain-first group without RBD (red dots) show initial reduced dopamine signal in the brain, while the impact on MIBG signal in the heart is affected later.
Brain First and Body First Parkinson's Disease - cohort 2 (The PACE-II cohort)
Introduction
Parkinson's disease (PD) is characterized by the loss of neurons in various regions of the brain and the peripheral nervous system, likely triggered by the accumulation of protein aggregates within neurons, particularly alpha-synuclein. The most severely impacted are the brain's neuromodulatory systems (such as the dopaminergic, noradrenergic, and cholinergic systems) and the autonomic nervous system in the body. However, emerging research indicates that neuronal damage in the brain and body does not occur simultaneously.
At our research centre, we propose an overarching hypothesis that PD manifests in two distinct subtypes: one where the initial pathology emerges in the brain or olfactory system (the brain-first subtype), and another where the pathology begins in the gastrointestinal tract and spreads via the autonomic nervous system to the brain (the body-first subtype). Consequently, patients with the body-first subtype may experience early neuronal loss in the autonomic nervous system, while those with the brain-first subtype may exhibit early neuronal loss in the brain's neuromodulatory systems.
This project aims to map the progression of neuronal loss in these vulnerable systems. In essence, we seek to determine the rate of degeneration within each system and for each subtype of PD. We will employ a comprehensive imaging protocol alongside thorough clinical assessments on newly diagnosed PD patients and follow them longitudinally to track changes over time.
Purpose
The main purpose of this study is to create progression maps of the vulnerable neuronal systems in the brain and body. Progression maps will be a powerful tool to:
- understand the complex presentation and evolution of PD symptoms
- confirm the existence of brain-first and body-first PD subtypes
- assess progression of the disease across multiple neuronal systems
- determine which imaging marker is optimal for assessing efficacy in neuroprotective trials, (depending on the PD subtype)
- to generate a unique, individual imaging signature that may serve as platform for future personalized treatments
Investigations
[18F]PE2I PET of the brain
Participants are injected with a small dose of [18F]PE2I. Thirty minutes later, a PET/CT of the head is performed. [18F]PE2I measures the dopamine system in the brain. The dopamine system is damaged in all patients with PD, but the degree of asymmetry and the progression of the damage might be different depending on the subtype of PD.
[18F]FEOBV PET of the brain
Participants are injected with a small dose of [18F]FEOBV. Three hours later, a PET/CT of the head is performed. [18F]FEOBV measures the cholinergic system in the brain. Degeneration of the cholinergic system seems to be closely related to cognitive decline but has also been associated with gait and balance difficulties.
MRI scanning of the brain
During magnetic resonance imaging (MRI), multiple sequences are conducted including a T1-weighted anatomical sequence, a short-duration T2-FLAIR sequence to rule out other brain diseases, and a special neuromelanin sequence to measure the amount of neuromelanin in the brainstem. We also acquire a special diffusion tensor image (DTI) to study microstructural damage. MRI is used to enhance the analysis of PET data and to measure certain brain systems that cannot be examined with PET scans.
[123I] scintigraphy of the heart
Participants are injected with a small dose of [123I]MIBG. The distribution of [123I]MIBG is visualized with a gamma camera 15 min (early image) and 210 minutes (late image) after injection. Together, these two images hold important information on the integrity of cardiac sympathetic nerve terminals. It is a powerful marker to distinguish brain-first from body-first subtypes of PD.
CT scanning of the abdomen
Every morning for 6 days, a capsule containing 10 very small plastic markers (radiopaque) is ingested. The plastic markers cannot be absorbed from the intestines. The number of retained markers in the intestines reflects the intestinal transit time. Many patients with PD display increased intestinal transit time, which is an objective marker of constipation.
Smell test
Participants are presented with 16 pens containing various familiar scents. From 4 options, they must identify the respective scent. The number of correctly identified scents is a measure of normal or impaired sense of smell. Patients with PD often develop impaired sense of smell at various stages of the disease.
Biological samples
We collect blood, urine, and saliva, which are analysed for protein aggregates, various inflammation markers and potential triggers of PD.
These samples are stored in a biobank for future research aimed at developing diagnostic markers, such as diagnosing PD through a blood test.
Blood pressure
Blood pressure is measured both in lying and standing positions to examine whether blood pressure drops more than usual (orthostatic hypotension). This is a symptom experienced by some patients with PD.
Motor symptoms
The motor symptoms are assessed using a standardized test (UPDRS), which evaluates the degree of slowness, stiffness, and tremors, among other factors. Motor function is also evaluated with more quantitative tests, such as moving the index finger back and forth between two points as many times as possible within one minute.
Pupillometry
A pupillometry measures pupil size and the reaction to a light stimulus. The pupil is controlled by the autonomic nervous system and may therefore be disturbed in some PD patients.
Other examinations
In addition to the above, a memory test is conducted along with various questionnaires covering symptoms commonly experienced with PD, such as constipation, sleep problems, etc.
Preliminary results
This project is still recruiting for the first publication. Preliminary results will be displayed here.
Brain first and body first Lewy body disease - cohort 3 (The PACE-III cohort)
We are currently working on the scientific protocol for our third and hitherto largest patient cohort – the PACE-III patient cohort.
We aim to start recruitment for this cohort in January 2026.