Research
Shine the searchlight of molecular biology on cells to decipher the underlying biology that drives malignancies!
Revolutionizing Prognostication
Leveraging Predictive Biomarkers for Precision Patient Stratification
Personalized treatment starts with understanding why patients differ.
Our work focuses on discovering molecular biomarkers that can predict how hematological cancers develop and respond to therapy. By integrating multi‑omics data with real‑world clinical information, we build smarter stratification tools that support risk‑adapted treatments and sharper clinical decisions.
Example in Action: Unraveling Lymphoma Pathogenesis and Prognosis
A powerful example of this effort is our work on B‑cell lymphomas, a group of cancers marked by striking biological heterogeneity. Each tumor carries its own molecular fingerprint, shaping how it behaves and how patients respond to treatment. To decode these differences, we apply large-scale molecular tools including mass‑spectrometry‑based proteomics, gene expression profiling, and immunohistochemistry.
By mapping the molecular features that shape disease progression, we aim to identify robust prognostic markers and sharpen risk stratification. These insights feed directly into our broader diagnostic pipeline, transforming advanced biological data into more precise, personalized clinical guidance.

Rethinking Diagnostics
Integrating AI-assisted Morphometrics for Enhanced Diagnostic Accuracy
Diagnostic precision remains a challenge especially in cases with atypical morphological or molecular features.
This subproject explores how cutting‑edge technologies such as artificial intelligence, digital pathology, and single‑cell profiling can be integrated to sharpen diagnostic accuracy. By revealing deeper layers of disease biology, our goal is to match patients more reliably to the right treatments and reduce diagnostic uncertainty.
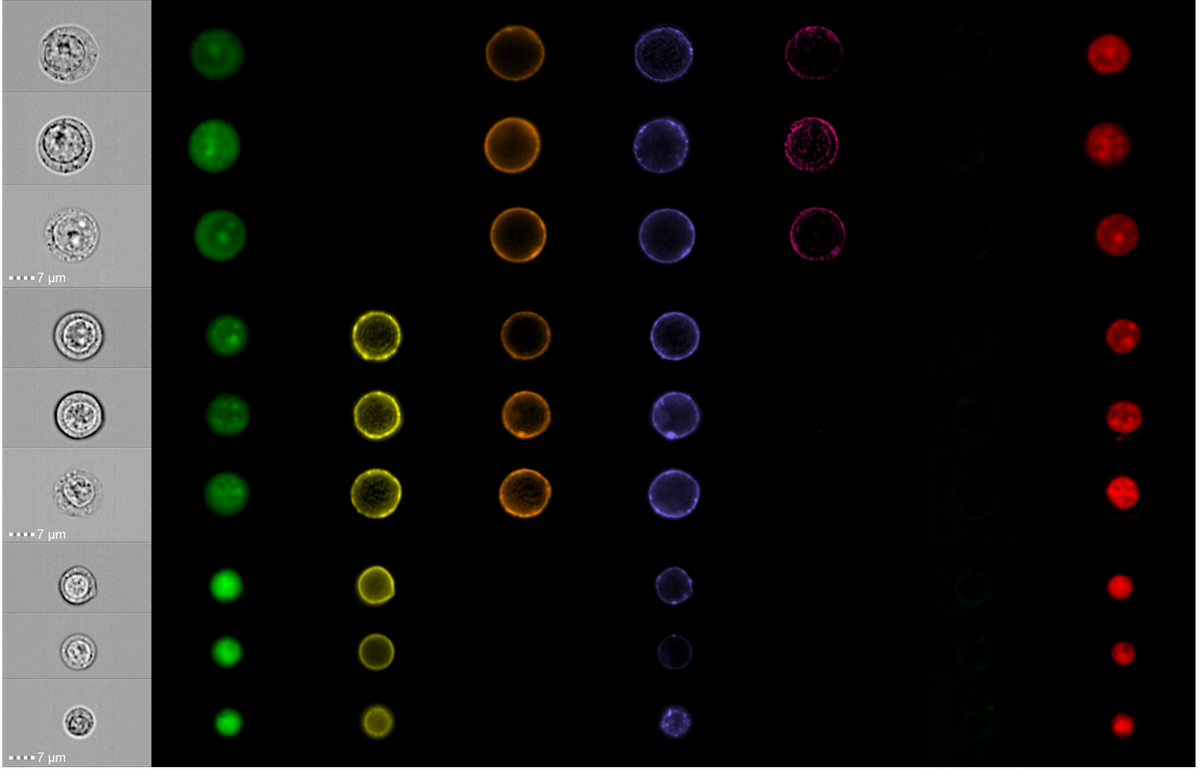
Example in Action: Morphometric Profiling at Scale
Traditional microscopy is essential but subtle changes in cell morphology can be hard to spot and vary between observers.
To push diagnostics forward, we apply imaging flow cytometry to diseases like MDS and AML. This technology merges high‑parameter flow cytometry with microscopy-level detail, capturing thousands of single cells with exceptional precision.
The result? Rich morphometric signatures that boost diagnostic accuracy and feed directly into our biomarker pipeline, driving more confident, individualized patient stratification.
Redefining Precision Oncology
Advancing Treatment by Paving the Way to Gene Therapy in Cancer
Gene therapy is transforming how we approach fusion oncogene (FO)–driven hematologic cancers.
This subproject aims to define the biological prerequisites and therapeutic windows needed to bring CRISPR‑Cas9 gene editing, delivered via lipid nanoparticles (LNPs), into relevant patient groups. By uncovering the mechanisms behind FO‑driven malignancies, we generate essential knowledge for translating gene‑based therapies into future clinical practice, unlocking major therapeutic potential.
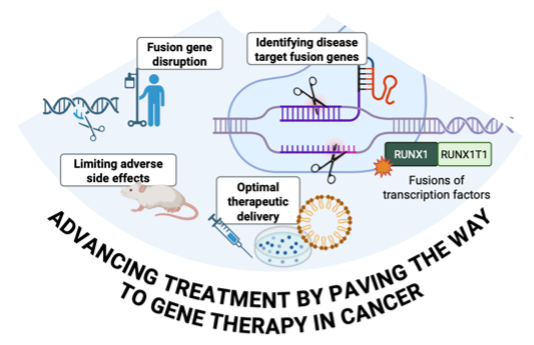
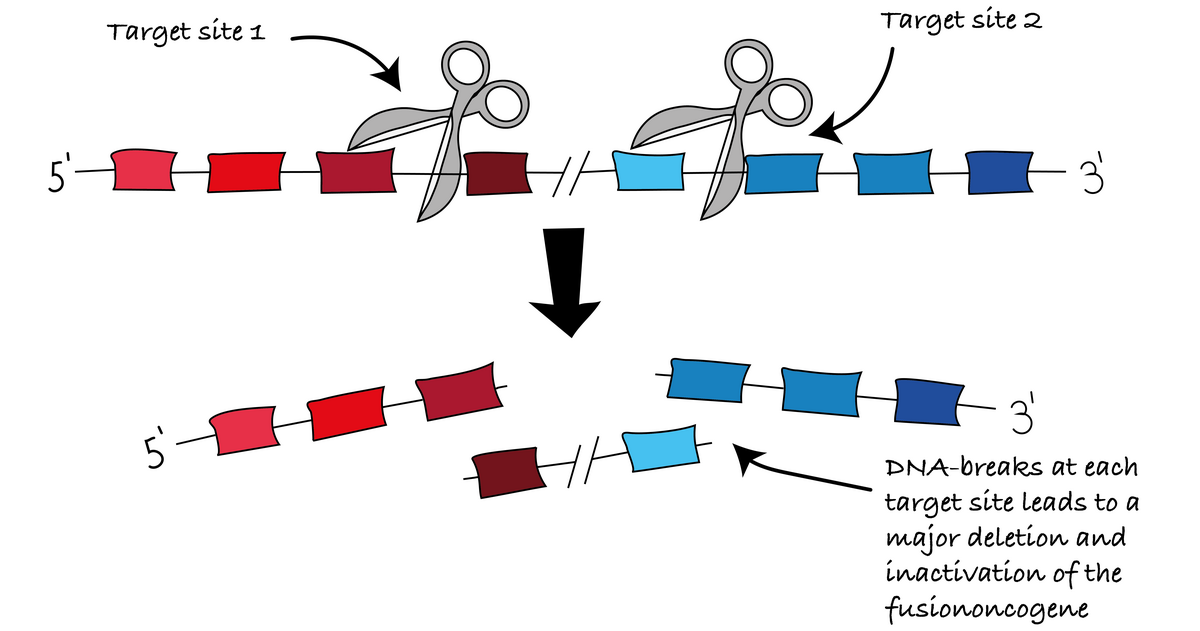
Example in Action: CRISPR for Treatment of Blood Cancers
A key example of this vision is our strategy to inactivate fusion oncogenes using CRISPR‑Cas9. We deploy two guide RNAs targeting intronic regions flanking the FO breakpoint. When CRISPR induces double‑strand DNA breaks at these positions, a large DNA deletion removes the fusion gene segment entirely, shutting down its oncogenic activity. Targeting conserved introns lowers the risk of unwanted genomic changes and supports an off‑the‑shelf therapeutic design.
Our current efforts span adult and pediatric acute myeloid leukemia, as well as bone and soft‑tissue sarcomas; diseases where innovative treatments are urgently needed.