Title:
COBRA NMIBC
Aim:
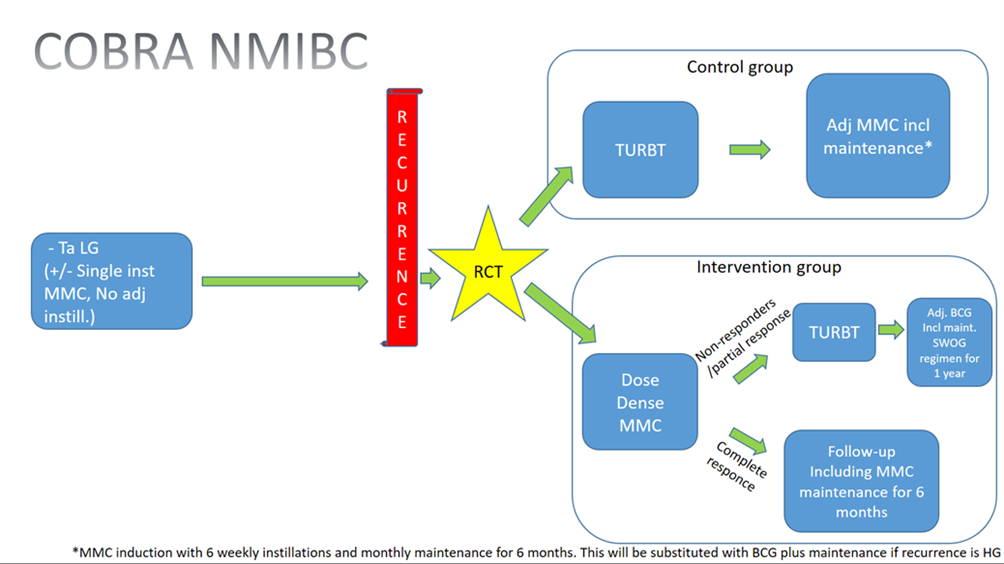
To assess if the efficacy of a dose-dense chemoablation with Mitomycin C (MMC) with adjuvant Bacillus Calmette–Guérin (BCG) in non-responding patients is superior regarding long-term effect compared to standard treatment with transurethral resection of bladder tumour (TURBT) and adjuvant intravesical instillation therapy in patients with recurrent Ta low-grade (LG) tumours.
Number of patients:
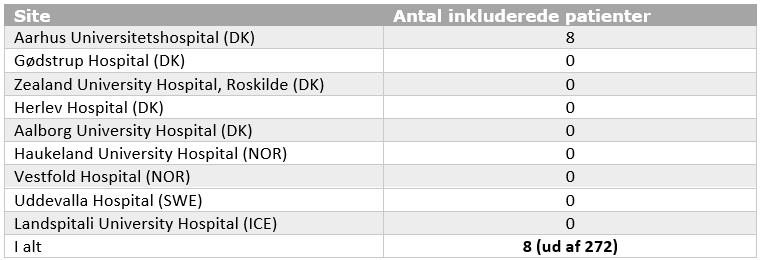
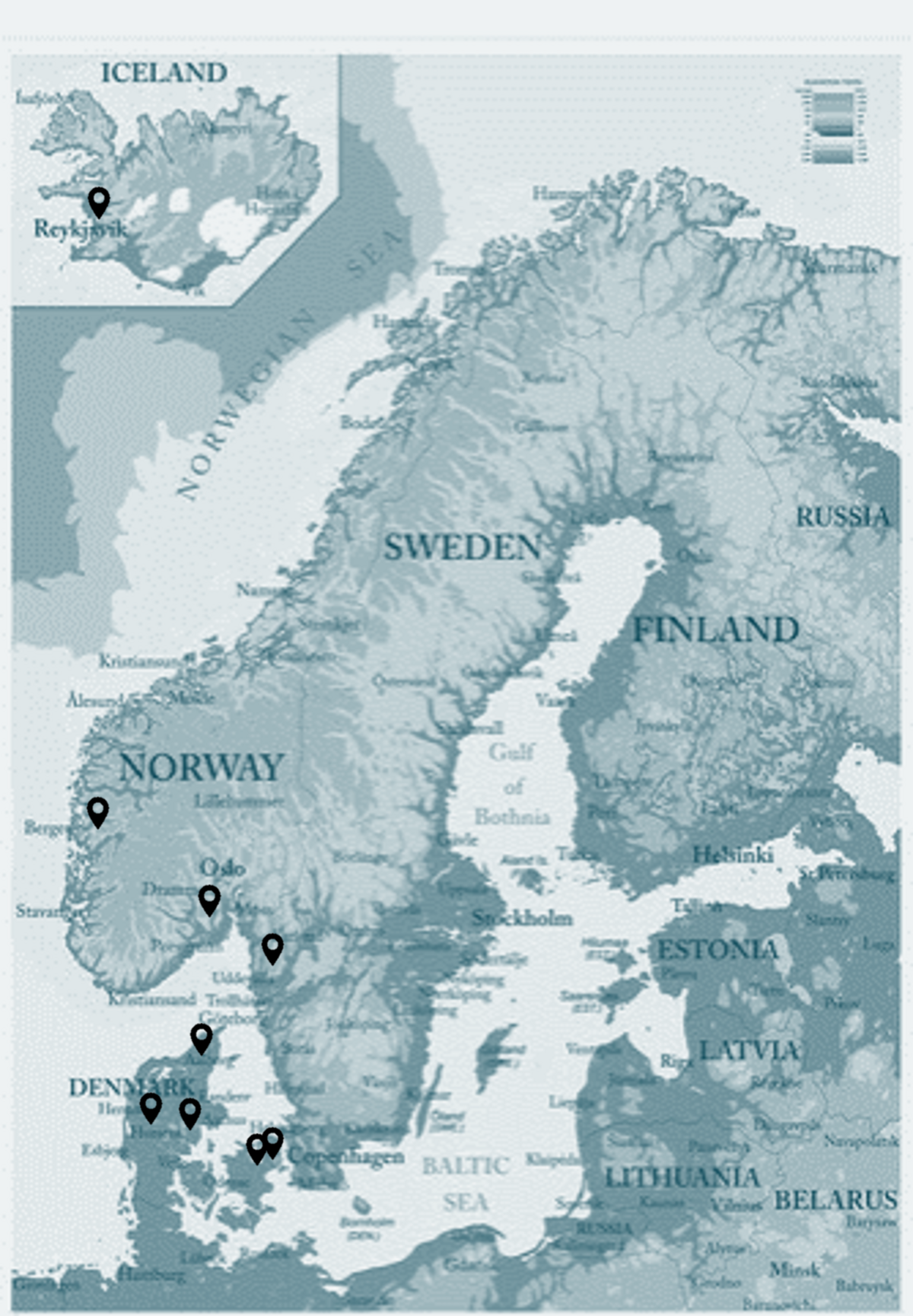
We expect to include 272 patients from 9 sites in the Nordic countries.
Methods:
The study will be conducted as an international multicentre randomised, controlled trial (RCT), where included participants are randomized between the intervention and control group. This trial design has been chosen to ensure the highest level of evidence (RCT). The international multicentre approach has been selected to ensure the highest degree of applicability of the results across countries and centres as previous studies have not had an international approach. The randomisation will be conducted 1:1.
Patient enrolment is expected to be ongoing for approximately 2 years in the following two arms:
Both groups follow the standard follow-up regimen according to European and national guidelines. Tumours with intermediate risk of recurrence are followed with a cystoscopy after three months, then once after nine months, then yearly for a minimum of 5 years if no recurrence. Tumours with high risk of recurrence are followed with cystoscopy and urine cytology every three months for two years, followed by cystoscopies twice a year until recurrence free for five years and then yearly.
End points are two-year recurrence-free survival (RFS), five-year RFS, and number of procedures (TURBTs or tumour fulguration) between groups.
Inclusion/exclusion criteria:
Inclusion criteria:
Exclusion criteria:
Status:
As of August 2025, 8 patients are enrolled from Aarhus University Hospital. Site initiation visits are planned in the fall of 2025.
Sites:
Participants will be included from 9 sites in Denmark, Sweden, Norway, and Iceland.
Clinicaltrial.gov:
NCT06781879